The U.S. Food and Drug Administration has accepted an Investigational New Drug application by physician-scientists at University of California San Diego School of Medicine to conduct the first U.S. clinical trial of an intravenously administered bacteriophage-based therapy. The planned trial will be conducted in collaboration with AmpliPhi Biosciences Corporation, a San Diego-based biotechnology company.
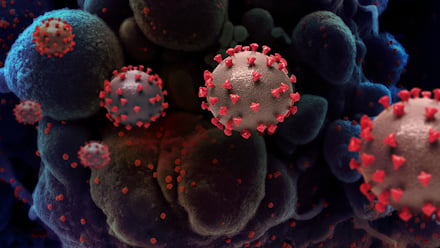
Bacteriophages, or phages, are viruses that specifically target and consume bacteria. They are ubiquitous, found wherever bacteria exist and were once considered a promising therapeutic tool. The advent of modern antibiotics in the 1930s redirected research interests, but with 10 million people estimated to die from drug-resistant “superbug” infections by 2050, they are getting a second look.
In 2017, a multi-institution effort led by UC San Diego scientists (and including AmpliPhi) successfully used an unprecedented phage therapy in a last-ditch, emergency effort to save a colleague dying from a multi-resistant bacterium.
That effort has been followed by a handful of singular cases, including the successful eradication of a years-long bacterial infection that allowed a patient to undergo a needed heart transplant.
Encouraged by the progress, UC San Diego Chancellor Pradeep Khosla last year awarded a three-year, $1.2 million grant to launch the Center for Innovative Phage Applications and Therapeutics (IPATH) in the UC San Diego School of Medicine, the first such center in North America. The clinical trial will be IPATH’s first.
The proposed Phase I/II clinical trial will test AB-SA01, an experimental bacteriophage combination for the treatment of participants with ventricular assist devices (VADs) infected by resistant Staphylococcus aureus (S. aureus). The trial will evaluate the safety, tolerability and efficacy of AB-SA01 bacteriophage therapy in combination with best available antibiotic therapies. There will be approximately 10 participants enrolled. They will be treated at UC San Diego Health and other leading teaching hospitals in the United States.
VADs are implantable mechanical pumps that help pump blood in patients with weakened hearts or heart failure. They are sometimes used as a transitional device for patients awaiting a heart transplant.
“There is a high, unmet need in patients with S. aureus VAD infections, which are typically very difficult to eradicate with conventional antibiotic therapy,” said the trial’s principal investigator, Saima Aslam, MD, associate professor of medicine and medical director of the Solid Organ Transplant Infectious Disease Service at UC San Diego Health.
“In 2018, our UC San Diego Health team treated a patient with a S. aureus VAD infection using AB-SA01 under AmpliPhi’s Expanded Access Program. This clinical trial builds on that foundational work and could provide a much-needed and promising treatment option for this life-threatening condition.”
Steffanie Strathdee, PhD, associate dean of global health sciences, Harold Simon Professor in the Department of Medicine and co-director of IPATH, said the planned trial is a natural progression from the first phage treatment at UC San Diego in 2017.
It was her husband, Tom Patterson, PhD, a professor in the Department of Psychiatry, who had been stricken by a seemingly untreatable bacterial infection while vacationing in Egypt. By the time Patterson was airlifted back to Thornton Pavilion, part of Jacobs Medical Center at UC San Diego Health, he was seriously ill and would soon slip into a coma.
“We’re excited to initiate IPATH’s first clinical trial,” said Strathdee. “And this is just the beginning. This collaboration is one of many we are undertaking to bring phage therapy into the 21st century to combat the global crisis of life-threatening antibiotic-resistant infections.”
- Visit the IPATH website
- Email IPATH at IPATH@ucsd.edu
Topics
Infectious Disease Clinical Trials