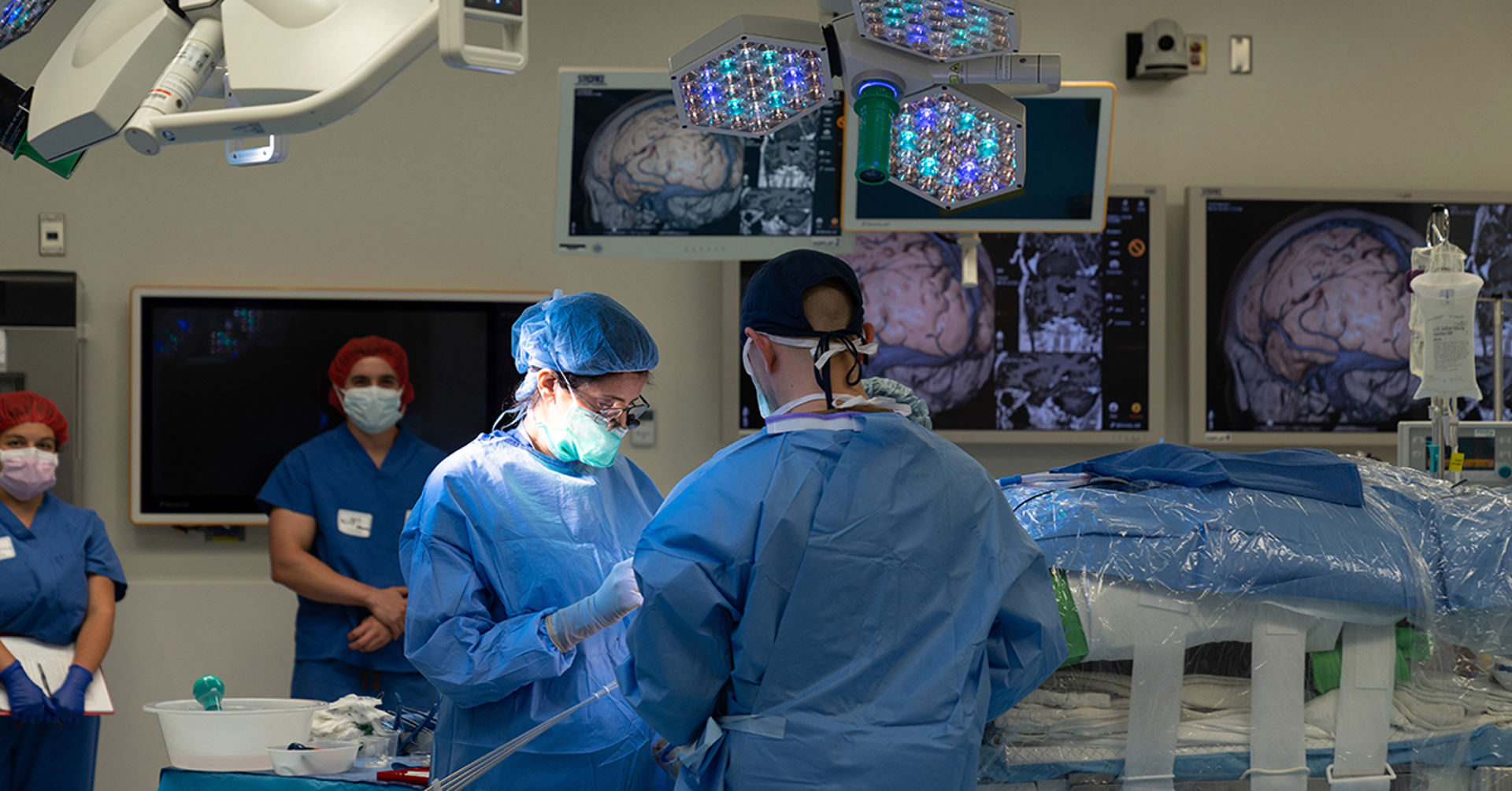
Sharona Ben-Haim, MD, performed the third ever experimental regenerative brain cell therapy for epilepsy earlier this month. The new procedure is part of a national clinical trial aimed at eliminating seizures, which could revolutionize the treatment of temporal lobe epilepsy.
In what could lead to a revolutionary advancement in the treatment of temporal lobe epilepsy, UC San Diego Health has become one of the first health systems in the country to inject regenerative cells into the brain to treat epileptic seizures.
Part of a national clinical trial, UC San Diego Health’s multidisciplinary team performed the third ever experimental regenerative brain cell therapy procedure earlier this month. UC San Diego Health is the only nationally designated Level 4 Adult Epilepsy Center in the region.
During the surgery, Sharona Ben-Haim, MD, associate professor of neurological surgery at University of California San Diego School of Medicine and surgical director of epilepsy at UC San Diego Health, made multiple injections of inhibitory brain cells into mapped out precision points of the patient’s brain under the bright lights of the operating room.
The cells, called interneurons, are derived from human stem cells. If successful, the first-ever regenerative human cell experimental therapy, NRTX-1001, could provide drug-resistant temporal lobe epilepsy patients with the first non-destructive option to potentially cure their seizures.
In between setting up multiple trajectory points on the patient’s brain prior to the cell insertion, Ben-Haim carefully studied the intra-operative magnetic resonance imaging scans that pinpoint her every move.
While digitally rotating the brain three-dimensionally on screen to inspect her work, Ben-Haim explained, "This experimental therapy offers us the potential to essentially restore the balance in the brain to be able to calm and ideally stop the seizures, while retaining the normal function of that part of the brain. Currently, we do not have a therapy that allows us to do that, so this is really exciting."
The clinical trial, sponsored by Neurona Therapeutics, is seeking to enroll 40 participants across the country to study the results of the implantation of the interneurons, which produce gamma-aminobutyric acid (GABA) — a neurotransmitter that blocks overactive impulses between nerve cells in the brain.
"In drug-resistant temporal lobe epilepsy, some of the normal brain cells in the temporal lobe have been damaged or are dead," said Jerry Shih, MD, professor of neurosciences at UC San Diego School of Medicine, neurologist and director of the Epilepsy Center at UC San Diego Health. "This experimental cell therapy implants healthy human brain cells into the damaged temporal lobe with the hope that those new cells will begin establishing connections in the patient’s brain, to ultimately make a healthier temporal lobe."
Epilepsy is the fourth most common neurological disorder in the U.S. after migraine, stroke and Alzheimer’s disease, according to the Centers for Disease Control and Prevention. An estimated 3.4 million Americans have epilepsy and approximately one-third of those individuals do not respond to anti-seizure medications.
Traditional seizure reduction therapies involve removing or laser-burning the parts of the brain where the seizures originate or implanting deep-brain electrodes to modulate seizure activities. This new experimental regenerative therapy could potentially treat multiple parts of the brain without tissue removal, offering new hope to drug-resistant epilepsy patients.
"This first-in-human clinical trial represents a paradigm shift in the way we treat this disease process, shifting from procedures that destroy bad tissue to procedures that repair the bad tissue," Shih said. "Our hope is that this procedure has such a high success rate and good tolerability that it becomes the standard of care for all drug-resistant focal epilepsies."
Back in the operating room, Ben-Haim meticulously inspected her work on her patient’s brain. He is the third person in the nation to undergo the procedure, which was initiated at SUNY Upstate Medical University in Syracuse, New York in June 2022, followed by Oregon Health Sciences University in Portland, Oregon in November 2022.
"These patients are willing to try an experimental procedure in this clinical trial to get control of their seizures, and I think they are incredibly brave," Ben-Haim said. "We are already seeing improvements in as early as one month. Our ultimate goal is to improve a patient’s long-term quality of life."
Patients who participate in the trial will be monitored regularly for two years after the procedure to study the effects of the implanted interneurons. Preliminary data reported in June demonstrates a more than 90% reduction in seizure frequency in the first and second patients at one year and seven months, respectively, post-treatment.
Shih, the principal investigator for UC San Diego Health’s involvement, said the study is the most complex clinical trial he’s served on in his career, spanning 25 years of conducting clinical trials at three leading academic institutions across the country.
"This study can only be conducted in an institution with a strong clinical and research infrastructure, which we are fortunate to have here at UC San Diego Health," Shih said.
He added that it required tremendous coordination among UC San Diego teams, including faculty and staff from neurosciences, neurosurgery, cellular regenerative medicine, radiology, neuropsychology and neuro critical care; California Institute for Regenerative Medicine Alpha Clinic at the Sanford Stem Cell Clinical Center; Advanced Cell Therapy Lab; Center for Multimodal Imaging and Genetics and the Consortium for Regenerative Medicine.
"We would not have been able to participate in this study without the active collaboration of all these integral groups. It truly takes a village."